The early years of the 19th century witnessed a rapid development in analytical chemistry-the art of distinguishing different chemical substances-and the consequent building up of a vast body of knowledge of the chemical and physical properties of both elements and compounds. Learn how the periodic table is organized See all videos for this article SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.The Hückel method, the extended Hückel method and the Pariser–Parr–Pople method, provide some quantitative theories. Molecular orbital diagrams provide simple qualitative LCAO treatment. These irreducible representations correspond to the symmetry of the orbitals involved. This reducible representation is decomposed into the sum of irreducible representations. The number of bonds that are unmoved is the character of that operation. Each operation in the point group is performed upon the molecule. The first step in this process is assigning a point group to the molecule. This is done by using the symmetry of the molecules and orbitals involved in bonding, and thus is sometimes called symmetry adapted linear combination (SALC).  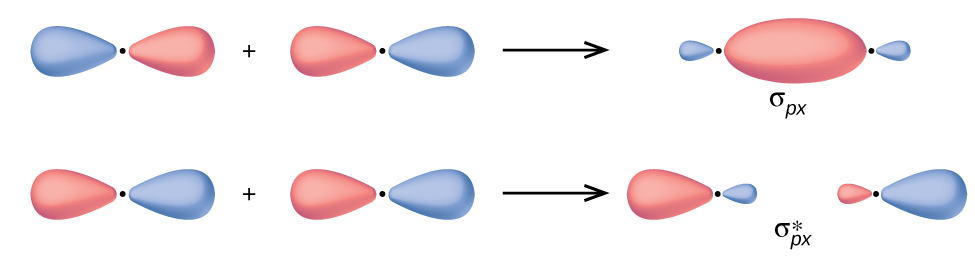
The required atomic orbital energies can come from calculations or directly from experiment via Koopmans' theorem. The graphs that are plotted to make this discussion clearer are called correlation diagrams. 
In this case, the shape of the molecular orbitals and their respective energies are deduced approximately from comparing the energies of the atomic orbitals of the individual atoms (or molecular fragments) and applying some recipes known as level repulsion and the like. However, since the development of computational chemistry, the LCAO method often refers not to an actual optimization of the wave function but to a qualitative discussion which is very useful for predicting and rationalizing results obtained via more modern methods. This quantitative approach is now known as the Hartree–Fock method. Slater-type orbitals but other choices are possible such as the Gaussian functions from standard basis sets or the pseudo-atomic orbitals from plane-wave pseudopotentials.īy minimizing the total energy of the system, an appropriate set of coefficients of the linear combinations is determined. The atomic orbitals used are typically those of hydrogen-like atoms since these are known analytically i.e. In either case the basis functions are usually also referred to as atomic orbitals (even though only in the former case this name seems to be adequate). The orbitals are thus expressed as linear combinations of basis functions, and the basis functions are single- electron functions which may or may not be centered on the nuclei of the component atoms of the molecule. The Hartree–Fock method is used to obtain the coefficients of the expansion. The coefficients are the weights of the contributions of the n atomic orbitals to the molecular orbital. Φ i = c 1 i χ 1 + c 2 i χ 2 + c 3 i χ 3 + ⋯ + c n i χ n, and r (numbered 1 to n) represents which atomic orbital is combined in the term. The expression (linear expansion) for the i th molecular orbital would be: In a sense, n atomic orbitals combine to form n molecular orbitals, which can be numbered i = 1 to n and which may not all be the same. The reducible representation of the bonding of water with C 2v symmetry.Īn initial assumption is that the number of molecular orbitals is equal to the number of atomic orbitals included in the linear expansion. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
